Evaluation of an oral telomerase activator for early age-related macular degeneration – a pilot study
Coad Thomas Dow1,2
Calvin B Harley3
1McPherson Eye Research Institute, University of Wisconsin-Madison, Madison, WI, USA; 2Chippewa Valley Eye Clinic, Eau Claire, Wisconsin, WI, USA; 3Independent Telomere Biology Consultant, Murphys, CA, USA
Abstract
Purpose—Telomere attrition and corresponding cellular senescence of the retinal pigment epithelium contribute to the changes of age-related macular degeneration. Activation of the enzyme telomerase can add telomeric DNA to retinal pigment epithelium chromosomal ends and has been proposed as a treatment for age-related macular degeneration. We report the use of a small molecule, oral telomerase activator (TA)-65 in early macular degeneration. This study, focusing on early macular degeneration, provides a model for the use of TAs in age-related disease.
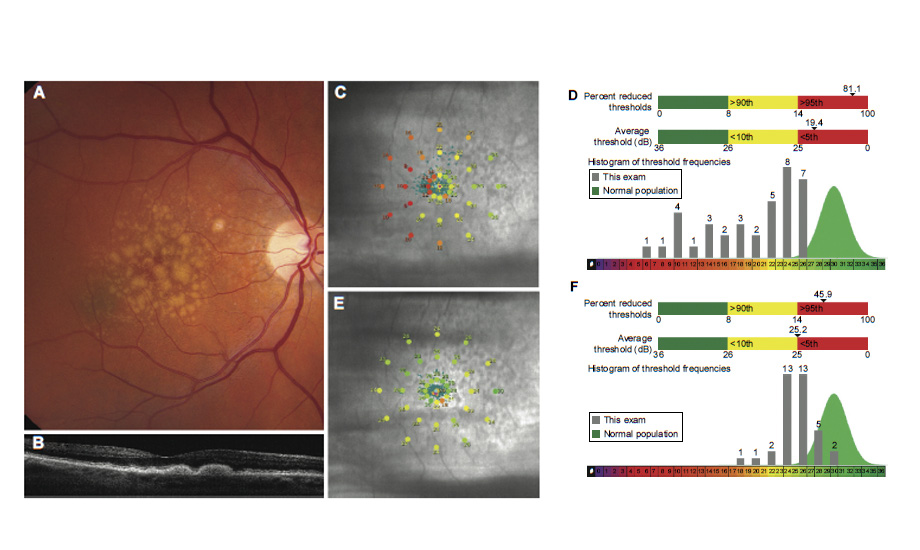
Method—Thirty-eight (38) patients were randomly assigned to a 1-year, double-blinded, placebo-controlled interventional study with arms for oral TA-65 or placebo. Macular functions via micro-perimetry were the primary measured outcomes.
Results—The macular function in the arm receiving the TA-65 showed significant improvement relative to the placebo control. The improvement was manifest at 6 months and was maintained at 1 year: macular threshold sensitivity (measured as average dB [logarithmic decibel scale of light attenuation]) improved 0.97 dB compared to placebo (P-value 0.02) and percent reduced thresholds lessened 8.2% compared to the placebo arm (P-value 0.04).
Conclusion—The oral TA significantly improved the macular function of treatment subjects compared to controls. Although this study was a pilot and a larger study is being planned, it is noteworthy in that it is, to our knowledge, the first randomized placebo-controlled study of a TA supplement.
Keywords
drusen, macular degeneration, micro-perimetry, senescence, telomerase activation, telomere (…)